Paper in Molecular Ecology Resources:
Beyond "living fossils": can comparative genomics finally reveal novelty?
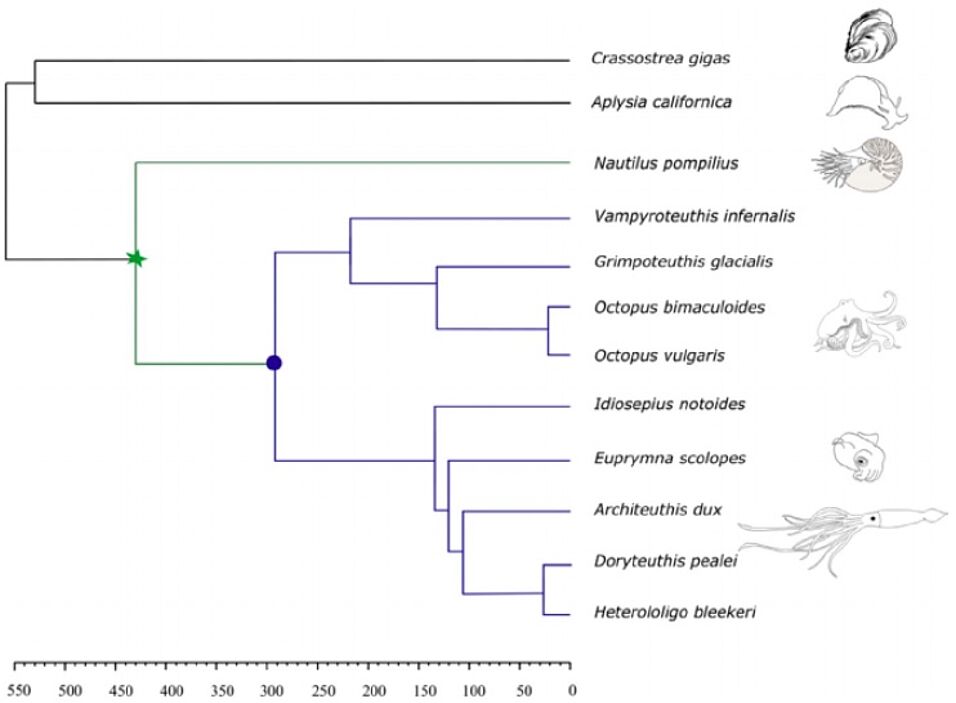
Cephalopods have recently moved into the research focus due to the growing number of sequenced genomes, molecular tools, and laboratory culture (Albertin & Simakov, 2020). Genome data now allows us to ask how the many known novelties of cephalopod morphology are reflected in their genomes and gene regulation. A crucial gap in this understanding has been the limited information for the Nautilus, the last survivor of a cephalopod lineage that diverged from the highly derived coleoid clade (octopus, squid, cuttlefish) around 400 million years ago. The publication of Nautilus genomes (in this issue of Molecular Ecology [Huang et al., 2021; Zhang et al., 2021]) will help us understand which genetic changes happened when, and ultimately how they contributed to cephalopod evolution.
Paper in Cell:
Whole-body integration of gene expression and single-cell morphology
Animal bodies are composed of cell types with unique expression programs that implement their distinct locations, shapes, structures, and functions. Based on these properties, cell types assemble into specific tissues and organs. To systematically explore the link between cell-type-specific gene expression and morphology, we registered an expression atlas to a whole-body electron microscopy volume of the nereid Platynereis dumerilii. Automated segmentation of cells and nuclei identifies major cell classes and establishes a link between gene activation, chromatin topography, and nuclear size. Clustering of segmented cells according to gene expression reveals spatially coherent tissues. In the brain, genetically defined groups of neurons match ganglionic nuclei with coherent projections. Besides interneurons, we uncover sensory-neurosecretory cells in the nereid mushroom bodies, which thus qualify as sensory organs. They furthermore resemble the vertebrate telencephalon by molecular anatomy. We provide an integrated browser as a Fiji plugin for remote exploration of all available multimodal datasets.